Does the Army really have a COVID-19 'super vaccine' and future variant killer?
Lots of hyped claims. No evidence just yet.
There are some wild headlines emerging on TV and in the corporate press about a U.S. Department of Defense-developed “super vaccine” for COVID-19, which is claimed to have the promise to work as a “pan-coronavirus” cure for any and all current and future variants.
Is the COVID-19 killer finally here? Your humble correspondent dug a little deeper into these claims.
The DOD’s Walter Reed Army Institute of Research (WRAIR) is doing a press tour for the preliminary results of a stage 1 trial for its Spike Ferritin Nanoparticle COVID-19 vaccine, or SpFN.
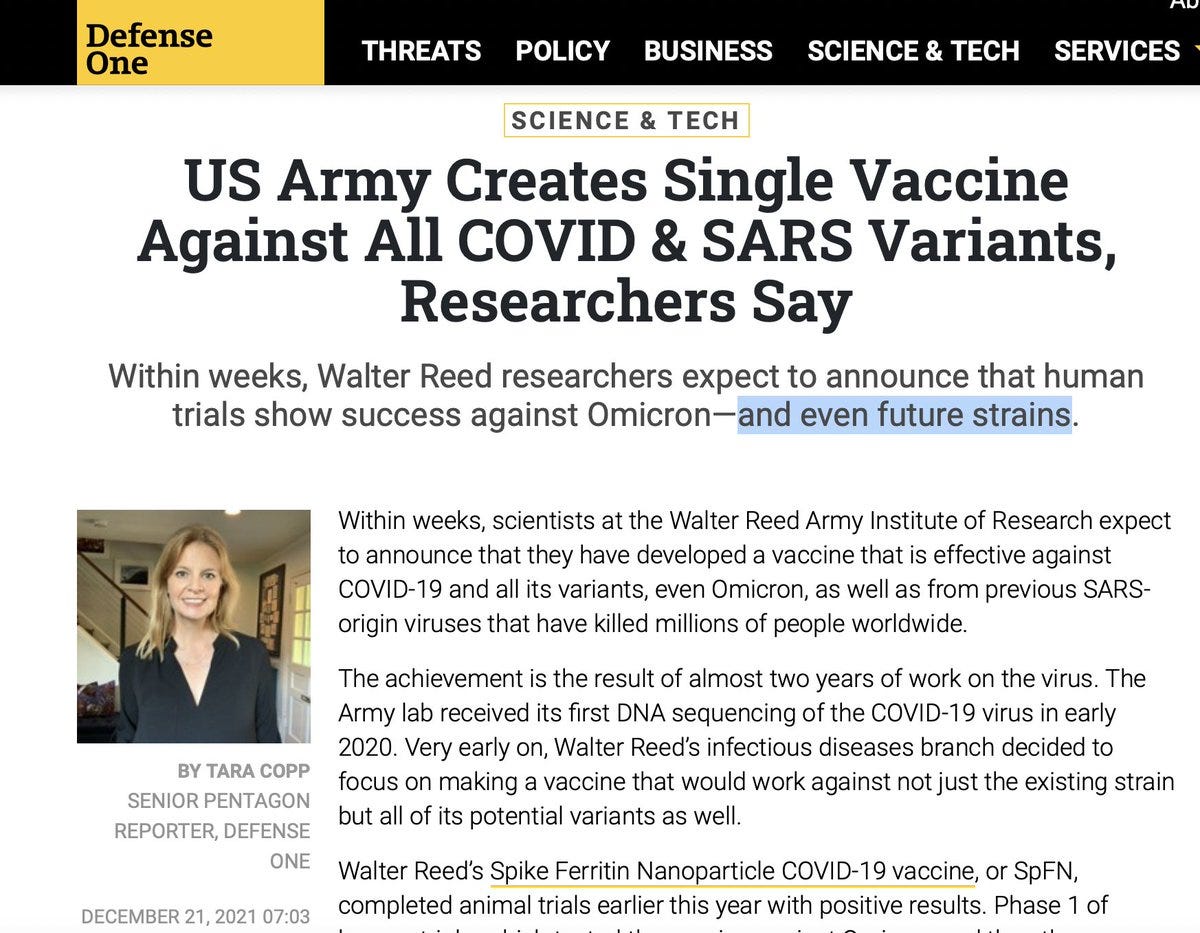
“Within weeks, scientists at the Walter Reed Army Institute of Research expect to announce that they have developed a vaccine that is effective against COVID-19 and all its variants, even Omicron, as well as from previous SARS-origin viruses that have killed millions of people worldwide.”
On December 16, the Army issued a press release about its nanoparticle COVID shot.
Kayvon Modjarrad, co-inventor of the vaccine and the U.S. Army lead for SpFN, said in a statement celebrating this development:
“Our strategy has been to develop a ‘pan-coronavirus’ vaccine technology that could potentially offer safe, effective and durable protection against multiple coronavirus strains and species.”
Modjarrad added:
“This vaccine stands out in the COVID-19 vaccine landscape. The repetitive and ordered display of the coronavirus spike protein on a multi-faced nanoparticle may stimulate immunity in such a way as to translate into significantly broader protection.”
The Army says that phase 1 human trials were completed at the beginning of this month, adding that their shots proved effective against the Omicron variant and “future strains.” It’s not clear how this Omicron claim is even possible, given that Omicron was first reportedly detected on November 24.
There are currently more than 60 nanoparticle-based COVID shots in pre clinical development, and 26 candidates have advanced into clinical trials.
Here’s what’s publicly know about the trial.
There’s 29 participants enrolled in the stage 1 human trials for the SpFN vaccine. All 29 individuals had to be deemed healthy, between the ages of 18-55, and having a BMI under 35 prior to being enrolled in the trial.
[95% of COVID-19 deaths occur in people who are over 50 years old. Additionally, the vast majority of people who have severe complications from COVID-19 are elderly and/or have several comorbidities. The most common underlying conditions for COVID-19 complications are all related to obesity. Therefore, the trial, while abiding by standard protocols, ruled out any participant who is realistically threatened by COVID-19.]
According to the trial information, the vaccine group is designed to have a 5 to 1 ratio to that of the placebo group. With only 29 of an expected 72 enrolled phase 1 trial participants, this means our control group sample size is tiny - a probable control group of 5 people. Yes, 5 whole human beings.
The stories that are populating the media, and the Army’s hype of a pan-coronavirus, cure-all, is not only unwarranted, but deeply suspect.
Meanwhile, just one day before the Army celebrated preclinical studies on its COVID cure-all shots, Dr. Anthony Fauci and his colleagues in Government Health published a paper titled, “Universal Coronavirus Vaccines — An Urgent Need.”
Check out our loyal sponsor:
TrustCapital is the #1 Bitcoin and precious metals IRA/401K platform in America. Sign up using my promo code DOSSIER and you will receive the benefits of tax-free investing and trading with ZERO MONTHLY FEES! Linked here and in the banner.







It will be SUPER and work against ALL Coronavirus variants ...especially future ones which we know almost nothing about! The operative word here is SUPER as in supercalifragilistic and Super Man, so we know its going to be UNSTOPPABLE .
LOL-The government under the last several administrations (elected and not elected members) has burned all its bridges with the people of the US. I don't trust them, and on the surface this claim seems patently absurd.
Nice Article.
I'd rename your article though.
I suggest "Bullshit on top of bullshit on top of bullshit with a little bullshit thrown in".
I discuss REAL AUTHENTIC BULLSHIT here:
https://timellison.substack.com/p/dont-you-hate-getting-ripped-off